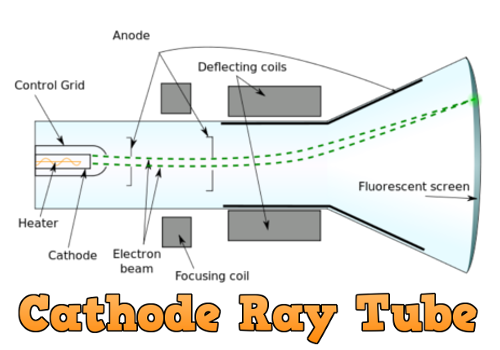 
Helped to further refine the atomic model.Led to the confirmation of the existence of the later named electron.Cathode rays had a large negative charge with a very small mass.As the charge-to-mass ratio could be calculated it demonstrated that cathode rays has measurable mass.Cathode rays were particles with a negative charge.As the magnetic field applies the force to move the cathode ray in a circular path, the magnetic field is applying a centripetal force and we can say:Ĭonclusions from Thomson’s Charge-to-Mass Ratio Experiment: The cathode ray then travels in an observable path of circular motion. The force on the cathode ray from the electric field and magnetic field can be equated to determine the velocity of the cathode ray:Īs the magnetic field is perpendicular to the cathode ray,ĭetermining the Charge-to-Mass Ratio of the Cathode RaysĪfter the velocity of the cathode ray has been determined, the electric field is turned off and the magnetic field is left on. The strength of the magnetic field can be adjusted so that the electric field and magnetic field forces are balanced and the cathode ray will pass through the CRT undeflected and strike the central position on the fluorescent screen. This forces the cathode ray to deflect back toward the central position. A magnetic field is then applied in such a way to oppose the force on the cathode ray created by the electric field. An electric field of strength E, is applied and the cathode rays deflect in the opposite direction to the electric field. They travel through the CRT to the end and strike the centre of the fluorescent screen. The cathode rays are made to accelerate from the cathode to the anode in a narrow beam. Determining the charge-to-mass ratio of the cathode raysĭetermining the Speed of the Cathode Rays.Determining the speed of the cathode rays. 
Thomson’s investigation consisted of two parts: He assumed that cathode rays were negatively charged particles and he used apparatus similar to that used by William Crookes to observe the properties of cathode rays. Thomson set out to measure the charge-to-mass ratio (q/m) for an electron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
